|
5/7/2023 0 Comments Density of copper 
Heavy materials should have density values greater than that of water. Light substances would float on water, so their density should be less than that of water. One way to do this is to mentally compare your answer against the density of water (1 gram per cubic centimeter). Density Cubes Set - Includes 6 Metals - Brass, Lead, Iron, Copper, Aluminum, Zinc - 0.4 (10mm) Sides - for use with Density, Specific Gravity Activities. With one free electron per atom in its metallic state, the electron density of copper can be calculated from its bulk density and its atomic mass. Finally, check to make sure your answer is reasonable. only cover up to 35 Amps, up to 0.4 inches of trace width, from 10 to 100 degrees C of temperature rise, and copper of 0.5 to 3 ounces per square foot. As an example of the microscopic view of Ohm's law, the parameters for copper will be examined. So, if you have four significant digits for mass but only three significant digits for volume, your density should be reported using three significant figures. Specific heat, or specific heat capacity, is a property related to internal energy that is very important in thermodynamics. The number of significant figures will be the same as the number in your least precise value. The other thing to watch is the number of significant figures in your answer. 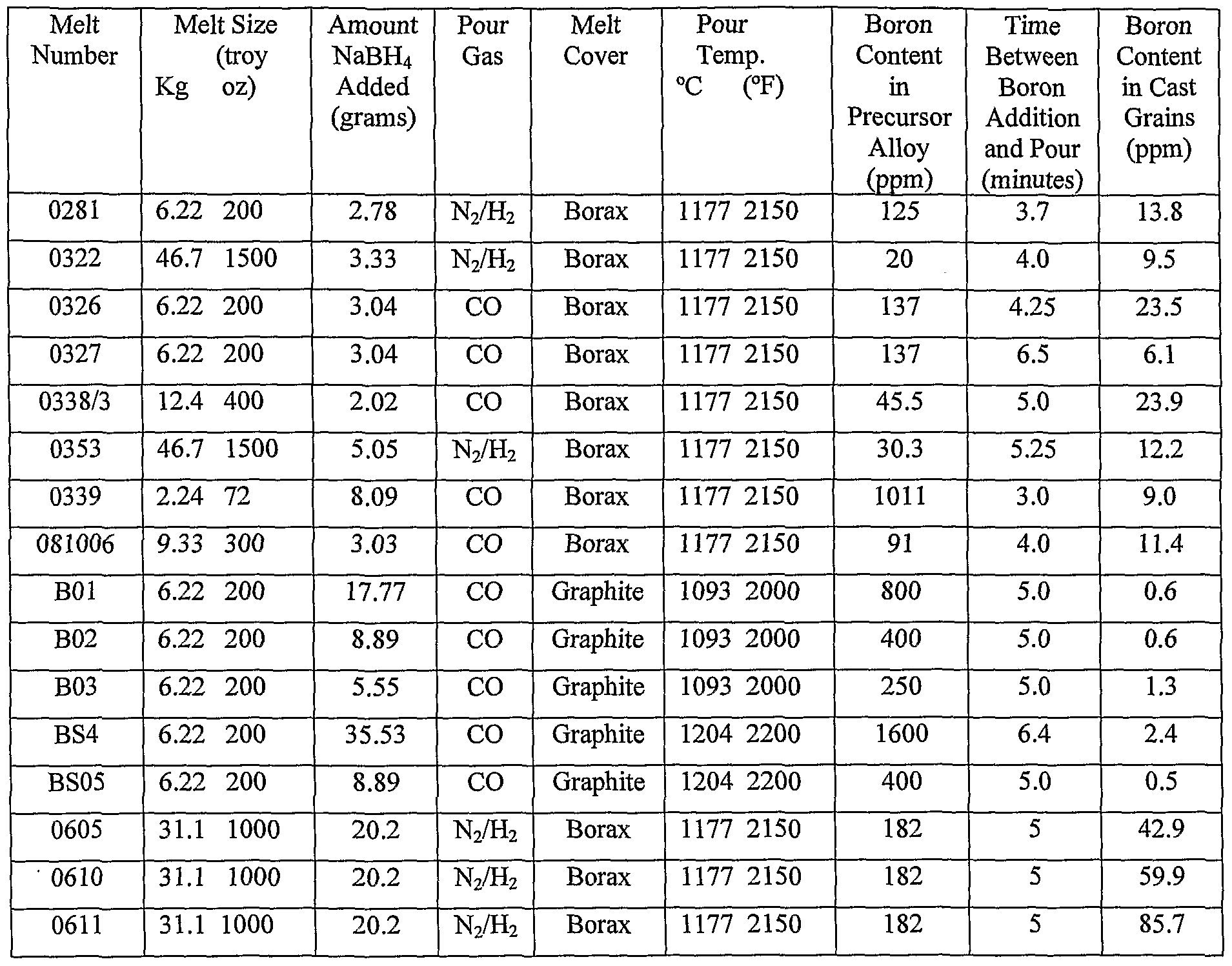
It's a good idea to be familiar with how to perform unit conversions when working on these problems. If you find this article useful then please like, share and comment. To see how density of materials is calculated, click here. It is a soft, malleable, and ductile metal with very high thermal and electrical conductivity. 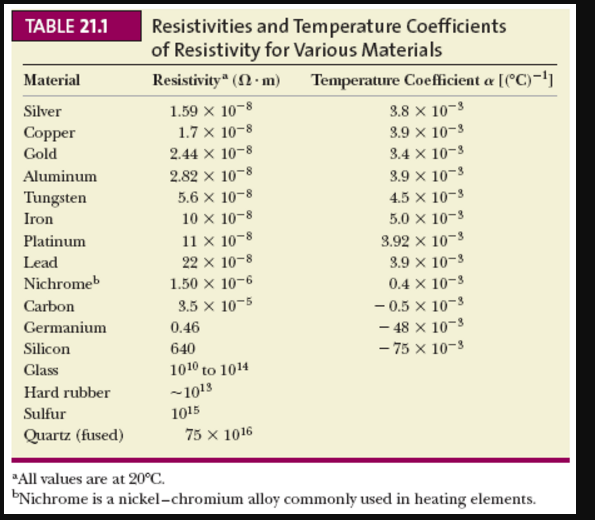
You may be asked to give an answer in different units than you're given. Material Properties Density of Metals Density of common metals By Paul Evans - 8 The typical density of common metals can be found listed in the table below. Copper is a chemical element with the symbol Cu (from Latin: cuprum) and atomic number 29. When you're asked to calculate density, make sure your final answer is given in units of mass (such as grams, ounces, pounds, kilograms) per volume (cubic centimeters, liters, gallons, milliliters). 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
